




 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
 |
 |
 |
|---|---|---|---|
The fundamental units are the meter, second, kilogram, and Coulomb. They were defined in 1793 as the "Standard International" (SI) units, or "MKS" units.
Quantity Unit Definition
Length Meter The Earth's circumference is 40 million meters
Time Second There are 86400 seconds in one Earth day
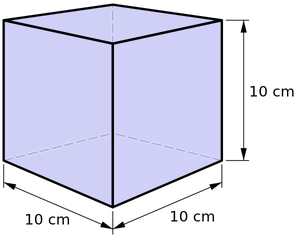
Mass Kilogram The mass of a cube of water 10 cm on a side is 1 kilogram
Charge Coulomb The force between two charges of one Coulomb each and
separated by 1 meter is 9 billion Newtons
 |
 |
 |
|---|---|---|
Density of water =1000 kg/meter = 1 g/cm Density of air = 1.2 kg/meter = .0012 g/cm
The fundamental units are length, mass, time, and charge, and all other units are derived from these.
Quanity Unit Composition Length meter X meter Mass kg M kg Time second T second Charge Coulomb C Coulomb Speed V = X/T Length / Time meter/second Acceleration A = V/T Speed / Time meter/second2 Momentum Q = M V Mass * Speed kg meter/second Force Newtons F = M A Mass * Acceleration kg meter/second2 Energy Joule E = F X Force * Distance kg meters2/second2 Power Watt P = E/T Energy / Time kg meters2/second3 Area S = X2 Length meters Volume Υ = X3 Length meters Density ρ = M/Υ Mass / Volume kg / meters2 Pressure Pascal Φ = F/S Force / Area Newtons/meter = Joules/meter Angular momentum L = M V X Momentum*Length kg meters/second Torque Γ = F X Force * Length kg meters/second Frequency Hertz f = 1/T 1 / Time 1/second
Meter = 3.281 feet
= 39.37 inches
Mile = 5280 feet (exact)
= 1609 meters
Foot = 12 inches (exact)
Inch = 25.4 mm (exact)
Minute = 60 seconds (exact)
Hour = 60 minutes (exact)
Day = 24 hours (exact)
Year = 365.25 days
Ton = 1000 kg (exact)
Kilogram = 1000 grams (exact)
= 2.205 pounds (pounds interpreted as mass)
Newton = .225 pounds (pounds interpreted as force)
Pound = 16 ounces (exact) (interpreted as mass)
= .454 kg
4.448 Newtons (Newtons interpreted as force)
Ounce = 28.35 grams (ounces interpreted as mass)
Meter/second = 2.24 miles/hour
Km/hour = .621 miles/hour
Miles/hour = 1.609 km/hour
Pascal = .0001450 pounds/inch2 (pounds interpreted as force)
Pound/inch2 = 6895 Pascals
Bar =101325 Pascals (Atmosphere pressure at sea level)
= 14.50 pounds/inch2 (pounds interpreted as force)
Earth gravity= 9.807 meters/second2
= 32.2 feet/second2
Standard sheet of paper = 11 x 8.5 inches = 27.94 x 21.59 cm
Meters Earth Earth Light travel
radii orbits time
(AU)
Nucleus 2⋅10
Atom 2⋅10
Green photon 5.5⋅10
Neuron .00002
Dime thickness .00135
Dime diameter .0178
Quarter diameter .024
Tennis ball diameter .067
Soccer ball diameter .22
Average person 1.78
Central Park width 800
Mount Everest 8848
Moon radius 1737000 .273
Mars radius 3390000 .532
Earth radius 6371000 1.0
Jupiter radius 6.991⋅107 10.9
Moon distance 3.844⋅108 60.3 .00257 1.5 seconds
Sun radius 6.957⋅108 109 .00474 2.3 seconds
Earth orbit 1.496⋅1011 23481 1.0 8 minutes
Jupiter orbit 5.2 40 minutes
Neptune orbit 30.1 3 days
Light year 9.461⋅1015 63241 1 year
Alpha Centauri 4.4 years Nearest star
Galaxy thickness 1000 years
Galaxy center 27200 years
Galaxy diameter 100000 years
Andromeda distance 2.5 million years
Virgo cluster distance 54 million years
Size of universe 14 billion years
 |
 |
 |
 |
 |
|
|---|---|---|---|---|---|
 |
|---|
 |
|---|
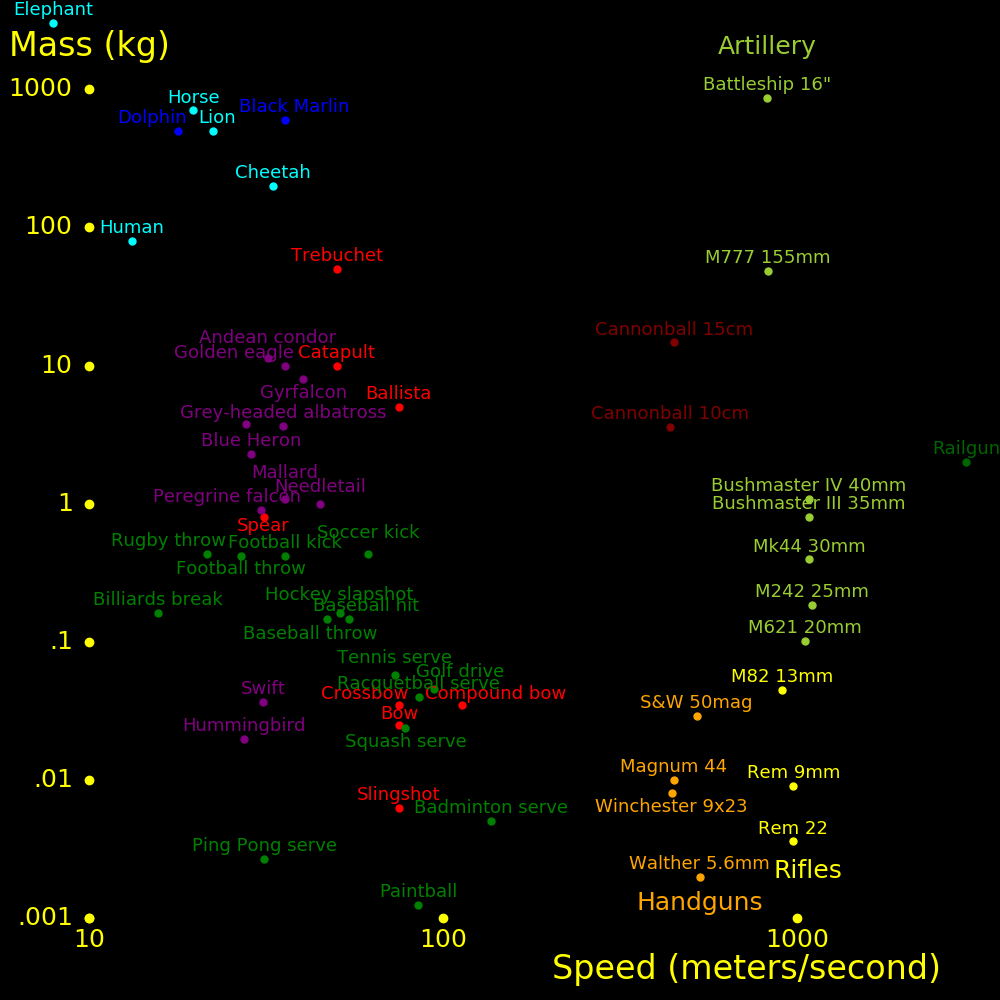
meters/second Mach
Walk 1.5
Running sprint 10
Cycling sprint 20
Cheetah 30 Fastest land animal
70 miles/hour 31
Baseball pitch 45 100 miles/hour
Human neuron 100
Sound at altitude 295 Speed of sound at altitude 10 km to 20 km
747 airplane 266 .9
Sound at sea level 340 1.0 At sea level and 15 degrees Celsius
F-35 Lightning 475 1.6 Stealth fighter
F-16 Falcon 590 2.0
Concorde 606 2.05
F-22 Raptor 670 2.3 Stealth fighter
F-15 Eagle 740 2.5
SR-71 Blackbird 980 3.3
Orbit speed 7800 26.4 Minimum speed to orbit the Earth
Escape speed 11200 38.0 Minimum speed to escape the Earth's gravity
Ion rocket 100000 Fastest spacecraft we can build
Fission rocket 107
Fusion rocket 107
Light 3⋅108 1020000
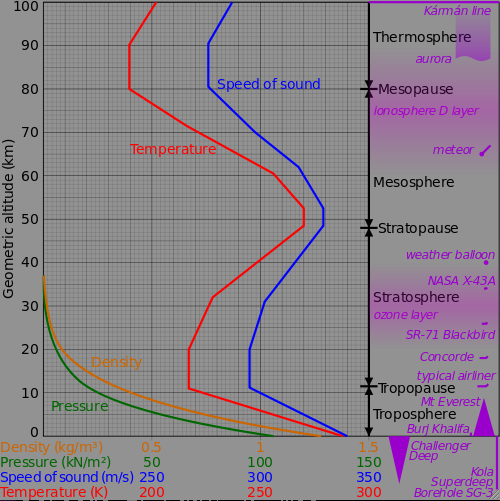
Aircraft typical fly at altitude 10 km to 20 km, where the speed of sound is
295 m/s. Mach 1 for aircraft is defined using this speed.
 |
 |
 |
|---|---|---|
kg Earth Solar
masses masses
Electron 9.109⋅10-31
Proton 1.673⋅10-27
Neutron 1.675⋅10-27
1 ounce .0283
Tennis ball .058
Soccer ball .44
1 pound .454
Typical human 70
Sumo wrestler 200
Ton 1000
Honda Civic 1200
Elephant 5000
Bradley tank 27000
Argentinosaurus 70000 Largest dinosaur
Blue whale 200000
Moon 7.35⋅1022 .0123
Mars 6.42⋅1023 .107
Earth 5.92⋅1024 1
Jupiter 1.90⋅1027 318 .00096
Sun 1.99⋅1030 330000 1.0
White dwarf max 2.9⋅1030 1.44
Milky Way black hole7.4⋅1036 4.2 million
Milky Way 2.5⋅1042 1.2 trillion
Andromeda 2.5⋅1042 1.2 trillion
M87 galaxy 10 trillion
Virgo galaxy cluster 1200 trillion
 |
|---|
 |
|---|
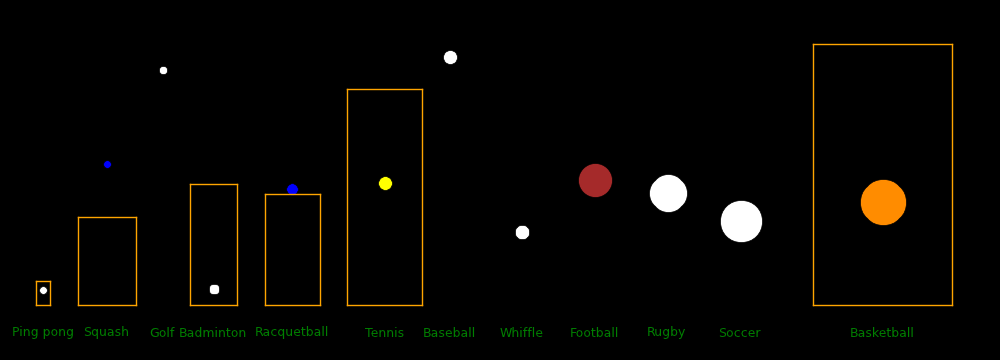
Ball Ball Court Court Ball Racquet Racquet Racquet Fastest Max Drag
diameter Mass length width density mass max length max width shot distance distance
mm gram meter meter gram/cm3 gram cm cm m/s meter meter
Ping pong 40 2.7 2.74 1.525 .081 70 31.2 1.8
Squash 40 24 9.75 6.4 .716 78.22 15.6
Golf 43 46 1.10 94.3 214.2 25.9
Paintball 1.25 85
Snooker 52.5 149 3.658 1.829
Badminton 54 5.1 13.4 5.18 .062 85 136.9 1.8
Racquetball 57 40 12.22 6.10 .413 85.4 12.8
Billiards 59 163 2.84 1.42 1.52 15.6 48.7
Tennis 67 58 23.77 8.23 .368 73.2 13.4
Cricket 72 160 80 .82 128.6 32.8 Throw 80ii meters from batter to home run boundary
158 Hit
Field hockey 73 160 91.4 55 .78
Baseball 74.5 146 122 .675 46.9 135.88 27.3 Throw Pitcher-batter distance = 19.4 m
54.14 177 Hit
Pickleball 74 24 13.41 6.10 .151 61.0 21.0
Hockey puck 76 163 61 26 1.44 51.0 25 mm thick
Whiffle 76 45 .196 8.1
Polo 82 130 274.3 146.3 .45
Croquet 92 454 1.11
Softball 97.1 188 .39 97.8 Throw
Softball 175.56 Hit
Football 178 420 91.44 48.76 .142 26.8 69.5 13.8 Throw
35.8 ~65 Placekick
90.5 Punt
Rhythmic gymn 190 400 12 12 .111
Rugby 191 435 100 70 .119 21.46 12.4 Throw
Volleyball 210 270 18 9 .056
Bowling 217 7260 18.29 1.05 1.36 160
Soccer 220 432 105 68 .078 35.84 9.3 Placekick
75.35 Punt
59.82 Throw-in
61.26 Throw
Basketball 239 624 28 15 .087 11.4
Disc ultimate 273 64 37 27.3 mm thick 18 meter end zonesDisc golf 300 200
Beach ball 610 120 .0011
Javelin 800 270 98.48
Discus 219 2000 74.08 44 mm thick
Hammer 102 7260 121.3 86.74
Shot put 125 7260 7.10 23.56
Cannonball 220 14000 7.9 945
Sumo 4.55
"Fastest shots" are world records.
 |
|---|
grams/cm2 $/kg Year of discovery
Magnesium 1.74 2.8 1808
Aluminum 2.70 1.7 1827
Titanium 4.51 10 1910
Zinc 7.14 2.0 1300
Manganese 7.21 2.3 1774
Iron 7.9 .3 -1200
Nickel 8.91 15 1751
Copper 8.96 6 -5000
Silver 10.49 640 Ancient
Lead 11.3 2 -6500
Tungsten 19.25 50 1783
Gold 19.30 43000 Ancient
Platinum 21.45 37000 1735
Osmium 22.59 12000 1803 Densest element
Air at Everest .0004 10 km altitude
Air at Denver .001 1 Mile altitude
Air at sea level .00127
Ice .92
Water 1.0
Rock 2.8
Earth 5.52
Moon 3.35
Mars 3.95
Europa 3.10
Ganymede 1.94
Callisto 1.83
Titan 1.88
Balsa .12
Corkwood .21
Cedar .32
Pine .37
Spruce, red .41
Oak, red .66
Hickory .81
Bamboo .85
Oak, live .98
Ironwood 1.1
Lignum Vitae 1.26
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
|---|
Mass Diameter Height Density Price/kg Copper Nickel Zinc Manganese
g mm mm g/cm3 $/kg fraction fraction fraction fraction
Penny 2.5 19.05 1.52 5.77 4.0 .025 .975
Nickel 5.000 21.21 1.95 7.26 10.0 .75 .25
Dime 2.268 17.91 1.35 4.62 44.1 .9167 .0833
Quarter 5.670 24.26 1.75 6.29 44.1 .9167 .0833
Half dollar 11.340 30.61 2.15 7.90 44.1 .9167 .0833
Dollar 8.100 26.5 2.00 7.53 123.5 .885 .02 .06 .035
Dollar bill 1.0 .11 .88 1000
The above objects are all to scale. The dimensions of a dollar bill are
155.956 mm * 66.294 mm * .11 mm.
Mass = M Diameter = D Height = H Volume = Vol = π H D2 / 4 Density = M / VolGold was the densest element known until the discovery of tungsten in 1783 and was hence valuable as an uncounterfeitable currency. Silver can be counterfeited with lead because lead is more dense and cheaper than silver.
The price of the metal in a penny is
Metal price = Penny mass * (Copper fraction * Copper price/kg + Zinc fraction * Zinc price/kg)
= .0025 kg * ( .025 6 $/kg .975 2 $/kg )
= .0052 $
For a penny made of pure copper the price of the metal is 1.5 cents.
A penny made of gold, silver, or zinc has a value of:
Price/Mass Price
$/kg $
Zinc 2 .005
Copper 6 .015
Silver 640 1.6
Gold 43000 108
For a 1 kg mass, Newton's law is:
Mass = M = 1 kg Gravitational acceleration = g = 9.8 m/s2 Gravitational force = F = M g = 9.8 Newtons = 2.203 poundsThe pound is a unit of force. 1 pound = 4.448 Newtons. In Earth gravity a mass of .454 kg produces a force of 1 pound.
Mass = M = .454 kg Gravitational acceleration = g = 9.8 m/s2 Gravitational force = F = M g = 4.448 Newtons = 1 pound
 |
|---|
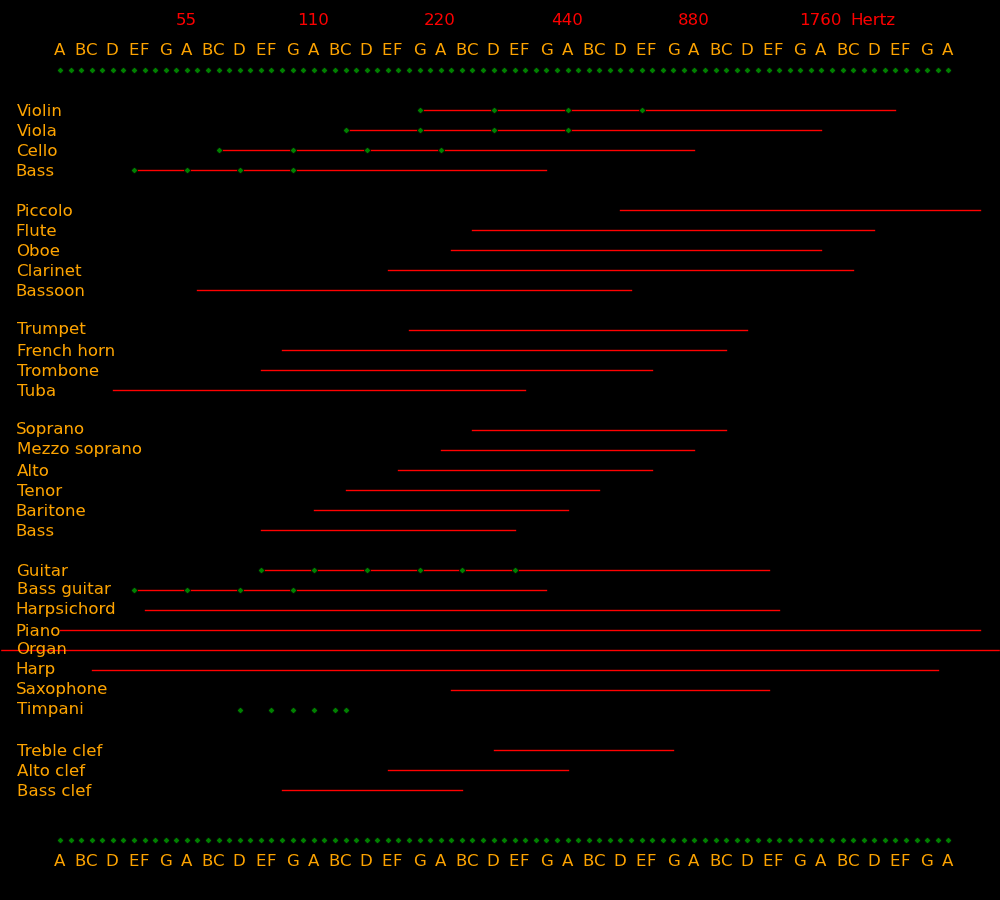
Frequency Note
(Hertz)
Whale songs 10
Human ear lower limit 20
Bass lowest note 41 E
Bass guitar lowest note 41 E
Cello lowest note 65 C
Bass singer lowest note 82 E
Viola & tenor lowest note 131 C
Violin & alto lowest note 196 G
Soprano lowest note 262 C
Violin D string 293 D
Violin A string 440 A
Violin E string 660 E
Human ear upper limit 20000
 |
 |
 |
 |
|---|---|---|---|
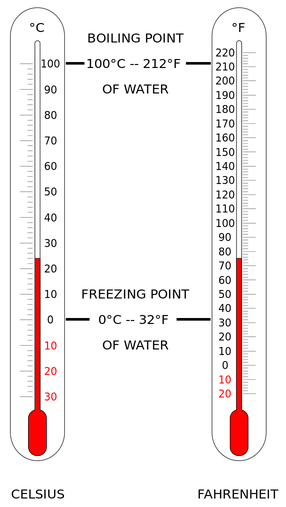
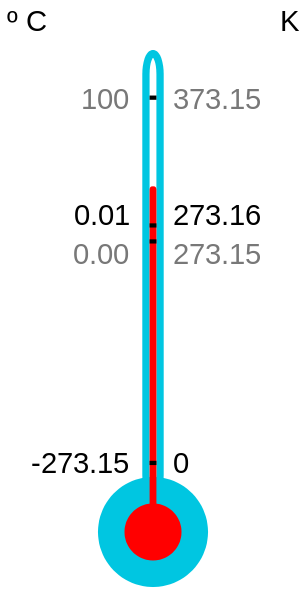
Kelvin Celsius Fahrenheit
Absolute zero 0 -273.2 -459.7
Water melting point 273.2 0 32
Room temperature 294 21 70
Human body temperature 310 37 98.6
Water boiling point 373.2 100 212
Kelvin
Absolute zero 0
Helium boiling point 4.2
Hydrogen boiling point 20.3
Triton 38
Pluto 44
Titania 70
Nitrogen boiling point 77.4
Oxygen boiling point 90.2
Titan 94
Europa 102
Hottest superconductor 135 HgBaCaCuO
Ceres 168
Mars 210
Water melting point 273.15
Earth average 288
Room temperature 293
Water boiling point 373.15
Venus 740
Wood fire 1170
Copper melting point 1358
Iron melting point 1811
Bunsen burner 1830
Tungsten melting point 3683 Highest melting point among metals
Earth's core 5650 Inner-core boundary
Sun's surface 5780
Solar core 13.6 million
Helium-4 fusion 200 million
Carbon-12 fusion 230 million
Color of a blackbody as a function of temperature (in Kelvin).
 |
|---|
Surface area = A Force = F Pressure = P = F / A (Pascals or Newtons/meter2 or Joules/meter3)
 |
|---|
Mass of the Earth's atmosphere = M = 5.15e18 kg
Surface area of the Earth = A = 5.10e14 m2
Gravitational constant = g = 9.8 m/s2
Pressure on Earth's surface = P = M g / A = 101000 Pascals
= 15 pounds/inch2
= 1 Bar
One bar is defined as the Earth's mean atmospheric pressure at sea level
Height Pressure Density
(km) (Bar) (kg/m3)
Sea level 0 1.00 1.225
Denver 1.6 .82 1.05 One mile
Everest 8.8 .31 .48
Airbus A380 13.1 .16 .26
F-22 Raptor 19.8 .056 .091
SR-71 Blackbird 25.9 .022 .034
Space station 400 .000009 .000016
Energy = E Joules Time = T seconds Power = P = E/T Watts Mass = M kilograms Energy/Mass = e = E/M Joules/kilogram Power/Mass = p = P/M Watts/kilogram
Energies in Joules.
1 food calorie 4200 1 Watt hour 3600 1 Watt * 3600 seconds Sprinting person 2560 80 kg moving at 8 m/s Battery, lithium, CR1216 330 Smallest button cell Battery, lithium, CR2032 3000 Most common button cell Battery, lithium-ion, AAAA 2300 Battery, lithium-ion, AAA 4700 Battery, lithium-ion, AA 9000 Battery, lithium-ion, A 47000 Battery, lithium-ion, B 58000 Battery, lithium-ion, C 67000 Battery, lithium-ion, D 107000 Battery, iPhone 7 (5 inch) 40000 Battery, Samsung S6 (5 inch) 52000 Battery, iPad mini (8 inch) 59000 Battery, iPad Pro (10 inch) 100000 Battery, iPad Pro (13 inch) 148000
Energies in MJoules:
1 kg of Lithium-ion battery .80 1 kg of TNT 4.2 1 kg of sugar 20 = 5000 Food Calories 1 kg of protein 20 = 5000 Food Calories 1 kg of alcohol 25 = 7000 Food Calories 1 kg of fat 38 = 9000 Food Calories 1 kg of gasoline 48 = 13000 Food Calories Tesla Model 3 battery 270 Fission bomb, uranium 8⋅107 = 20 kilotons of TNT Fusion bomb 8⋅1010 = 20 megatons of TNT World energy used in 1 year 6⋅1014
Forms of energy:
Distance = X meters Force = F Newtons Mass = M kg Velocity = V meters/second Gravity constant = g = 9.8 meters/second2 Pressure = P Pascals Volume = U meters3 Mechanical energy= Ew = F X Joules Gravity energy = Eg = MgX Joules (X = height above ground) Kinetic energy = Ek = ½MV2 Joules Pressure energy = Ep = P U Joules
Watts
Human cell 10-12
iPhone 7, standby .05
iPhone 7, audio .3
iPhone 7, video .9
iPhone 7, talk .9
iPad Pro 10 inch, idle 3
Human brain 20
Incandescent Light bulb 60
Human at rest 100
Unstrenuous cycling 200
1 horsepower 746
Strenuous cycling 600
Maximum human power 1600
World power per person 2500
Tesla S Ludicrous 397000 532 horsepower
Wind turbine 1⋅106
Blue whale 2.5⋅106
Boeing 747 1.4⋅108
Hoover Dam 2.1⋅109
U.S. power consumption 3.4⋅1012
World power consumption 1.5⋅1013
Earth geologic heat 4.4⋅1013
World photosynthesis 7.5⋅1013
Earth solar power 1.7⋅1017 Total solar power falling on the Earth
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
 |
 |
|---|---|---|
 |
 |
 |
 |
 |
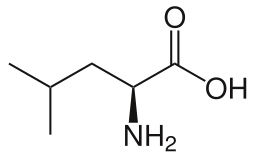 |
|---|---|---|---|---|---|
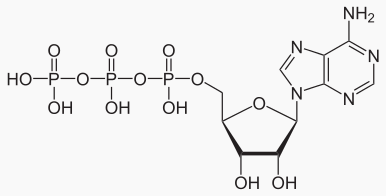 |
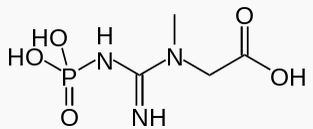 |
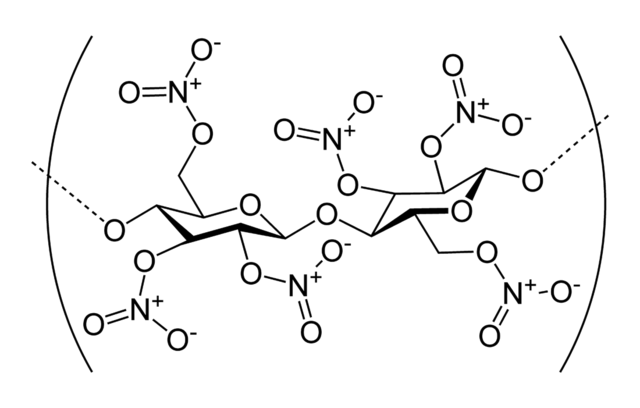 |
 |
 |
|---|---|---|---|---|
 |
 |
|---|---|
 |
 |
 |
 |
 |
 |
 |
|
|---|---|---|---|---|---|---|---|
 |
|---|
Energies and powers are for lithium batteries, which have a voltage of 3.7 Volts. The "ID #" is often used instead of cell size.
Cell Energy Power Current Mass Diameter Length Charge Price ID # size kJoule Watt Ampere gram mm mm AmpHour $ D 107 220 60 138 32 67 8.0 13 32650 C 67 220 60 92 26 50 5.0 8 26650, 25500 B 58 160 45 72 22 60 4.5 5 21700, 20700 A 47 110 30 49 18 50 3.5 3 18650 AA 9 22 6 15 14 53 .70 1 14500 AAA 4.7 11 3 7.6 10 44 .35 .5 10440 AAAA 2.3 6 1.5 3.8 8 42 .17 .25 75400 CR2032 3. Most common button cell CR1216 .33 Smallest button cell Apple Watch 4 4.0 .29 iPhoneXR 6" 41 2.94 Machine = .194 kg iPhoneXSM 6" 44 3.17 Machine = .208 kg iPhoneXS 6" 36 2.66 Machine = .177 kg iPhone8+ 6" 27 2.79 Machine = .202 kg iPhone8 5" 25 1.82 Machine = .148 kg iPhone7+ 6" 40 2.90 Machine = .188 kg iPhone7 5" 27 1.96 Machine = .138 kg iPad Mini 8" 70 5.12 Machine = .30 kg iPad Pro 10" 111 8.13 Machine = .47 kg Mac Air 11" 137 Machine = 1.08 kg Mac Air 13" 194 Machine = 1.34 kg MacBook 12" 149 Machine = .92 kg Mac Pro 13" 209 Machine = 1.37 kg Mac Pro 15" 301 Machine = 1.83 kg
Energy Power Lifetime
kJoule Watts hours
iPhone 8 5" 25 .50 14
iPhone 8+ 6" 27 .54 14
iPad Mini 8" 70 1.9 10
iPad Pro 10" 111 3.1 10
Mac Air 11" 137 3.8 10
Mac Air 13" 194 5.4 10
Mac Pro 13" 209 5.8 10
Mac Pro 15" 301 8.4 10
Meters/second2
Ceres gravity .27
Europa gravity 1.31
Titan gravity 1.35
Moon gravity 1.62
Mars gravity 3.8
Venus gravity 8.87
Earth gravity 9.8
Bugatti Veyron 15.2 0 to 100 km/h in 2.4 seconds
Red out 30 Max long-term acceleration in the direction of blood rushing to your head
Blackout 50 Max long-term acceleration while sitting
Formula-1 car 50 High-speed breaking and cornering with a downforce wing
Blackout with g suit 90 Max long-term acceleration while sitting with a g-suit
Max long-term (front) 120 Max long-term acceleration while lying on one's front
Max long-term (back) 170 Max long-term acceleration while lying on one's back
Max short-term 500 Max short-term acceleration
Bullet 310000 9x19 Parabellum handgun, average acceleration along the barrel
terameter = Tm = 10 meters gigameter = Bm = 10 meters megameter = Mm = 10 meters kilometer = km = 10 meters meter = m = 10 meters centimeter = cm = 10 meters millimeter = mm = 10 meters micrometer = μm = 10 meters nanometer = nm = 10 meters picometer = pm = 10 meters femtometer = fm = 10 meters 1 million kg = 1 Mkg 1 million dollars = 1 M$
Examples of scientific notation.
1 = 100 = e0
10 = 101 = e1
100 = 102 = e2
123 = 1.23⋅102 = 1.23e2
.123 = 1.23⋅10-1 = 1.23e-1
11000 * .012 = 1.1⋅104 * 1.2⋅10-2 = 1.32⋅102 = 132
The abbreviation "e" for "10^" comes from Fortran
and is standard in all programming languages.
A measurement consists of a quantity and an estimated error. For example, you might measure the length of a room to be
Length = 6.35 ± .02 meters"6.35" is the measurement and ".02" is the estimated error.
Care should be taken to use an appropriate number of digits. For example,
Length = 6.3 ± .02 meters Not enough digits in the measured quantity Length = 6.34 ± .02 meters Minimum number of digits to state the measured quantity Length = 6.342 ± .02 meters It is wise to to include an extra digit Length = 6.3421 ± .02 meters Too many digits. The last digit is unnecessary.The fractional error is defined as
Fractional error = Error / Measured quanitity
= .02 / 6.34
= .0032
Rounding:
6.3424 → 6.342 6.3425 → 6.342 6.3426 → 6.343If the last digit is even then round down, and if odd then round up. This prevents bias in rounding. For example:
6.05 → 6.0 6.15 → 6.2 6.25 → 6.2 6.35 → 6.4 6.45 → 6.4 6.55 → 6.6 6.65 → 6.6 6.75 → 6.8 6.85 → 6.8 6.95 → 7.0
1 mile = 1609 meters
1 hour = 3600 seconds
1609 meters 1 hour
1 mile/hour = 1 mile/hour * ----------- * ------------ = .447 meters/second
1 mile 3600 seconds
If you have data that is not in SI units, then the safest procedure is to convert everything to SI units do the calculation. You can't go wrong with this. For example, if a car moving at 70 mph travels for 2 hours, how far does it go?
Speed of a car = V = 70 mph = 31.3 meters/second Time traveled = T = 2 hours = 7200 seconds Distance traveled = X = V T = 140 miles = 225360 metersOne first converts 70 mph and 2 hours to SI units, then apply X=VT to arrive at X=225360 meters, and then convert this to mph.
Alternatively, you can do the calculation in non-SI units but care must be taken to make sure the units are consistent.
The logarithm is the inverse of the exponential function.
10-2 = .01 log10 .01 = -2 10-1 = .1 log10 .1 = -1 100 = 1 log10 1 = 0 101 = 10 log10 10 = 1 102 =100 log10100 = 2 10log10x = log10 10x = x
Suppose you measure the power exerted in climbing a set of stairs.
Height of stairs = H = 20 meters Time to climb stairs = T = 10 seconds Vertical speed = V = H/T = 2 meters/second Mass of climber = M = 100 kg Gravity constant = g = 10 meters/second2 Gravity energy = E = MgH = 20000 Joules Power = P = E/T = 2000 WattsThere is a row for each variable and there are 5 columns showing the properties of each variable. The columns are:
*) Description of the variable
*) Symbol for the variable
*) Units-style equation
*) Numerical example
*) Units
Most units-style calculations can be done with this recipe.
If a smartphone is being used to play League of Legends, typical values for the lithium battery are
Energy = E = = 20000 Joules (Typical smartphone battery energy) Lifetime = T = = 14400 seconds (While playing League of Legends) Power = P = E/T = 1.39 Watts Mass = M = .027 kg Energy/Mass = e = E/M = .75 MJoules/kg (Typical value for lithium batteries) Power/Mass = p = P/M = 52 Watts/kgThe maximum power/mass that a lithium battery is capable of producing is 750 Watts/kg.
It takes .7 kg of rice to feed one human for one day.
Energy in 1 Calorie = 4200 Joules Time = T =86400 seconds = 1 day Food energy in one day = E = 10.5 MJoules = 2500 Calories Power = P = E/T = 121.5 Watts Rice mass = M = .7 kg Rice energy/mass = e = E/M = 15 MJoules/kgIn this example we added a column for non-SI units (days and Calories), which have to be converted to SI units.
The price of electricity is 5 cents per kiloWatt hour.
Power = P = 1000 Watts = 1 kiloWatt Time = T = 3600 seconds = 1 hour Energy = E = PT = 3.6 MJoules Price = C = E/c = .05 $ Energy/$ = c = 72 MJoules/$
The kinetic energy of an object in orbit is 32 MJoules/kg, and it costs .44 dollars for this much energy in electricity. The real launch cost is 2000 dollars/kilogram.
Orbital velocity = V =8000 meters/second Mass = M = 1 kg Kinetic energy = E = ½MV2 = 32 MJoules Electricity energy/$ = c = 72 MJoules/$ Electricity cost = C = E/c = .44 $
Many things are best understood by constructing a 2D table of numbers. For example, suppose you're wondering how to compare the alcohol content of a 6 pack of beer, a bottle of wine, and a bottle of Scotch.
A standard bottle of beer is 12 ounces, has an alcohol fraction of .05, and the volume of alcohol is .6 ounces. For other drinks,
Alcohol Drink Alcohol Alcohol
fraction volume volume volume
(oz) (oz) (Beers)
Beer (12 oz) .05 12 .6 1
Wine glass .13 4.6 .6 1
Scotch shot .40 1.5 .6 1
Beer pitcher .05 64 3.2 5.3
Beer keg .05 1984 99.2 165.3
Wine bottle .13 25.4 3.3 5.5
Scotch bottle .40 25.4 10.1 16.9
Distilled ethanol .95 25.4 24.1 40.2
The "alcohol volume" column provides a comparison between different drinks.
In a 2D table, columns are units and rows are items. Each column has a label and a unit. For example in the alcohol table, one of the columns is labeled "alcohol volume" and it has units of ounces. Monospace fonts are helpful for aligning columns.
Numbers in columns are easier to compare than numbers in rows, and this is why units are aligned as columns rather than rows. Most often it is units that are being compared.
We define a strategic unit, the "beer", which is the volume of alcohol in a standard beer. 1 beer = .6 ounces. In the table's last column we show the alcohol volume in units of beers.
We can expand the table to show the cost of each form of alcohol.
Alcohol Volume Alcohol Alcohol $ $/Beer
fraction (oz) (oz) (Beers)
Beer (12 oz) .05 12 .6 1 .67 .67 Budweiser
Wine glass .13 4.6 .6 1 8 8.0 Napa Valley
Scotch shot .40 1.5 .6 1 8 8.0 Laphroaig
Beer pitcher .05 64 3.2 5.3 16 3.0 Budweiser
Beer keg .05 1984 99.2 165.3 100 .60 Budweiser
Wine bottle .13 25.4 3.3 5.5 3 .55 Charles Shaw
Vodka bottle .40 25.4 10.1 16.9 15 .89 Smirnoff
Scotch bottle .40 25.4 10.1 16.9 50 3.0 Laphroaig
Distilled ethanol .95 25.4 24.1 40.2 15 .37 Everclear
A table is usually accompanied by equations showing how the columns are related.
Volume of the drink = V Fraction of alcohol = F Volume of alcohol = Valc = F V Volume of one beer = Vbeer = 12 ounces Alcohol fraction of beer = Fbeer = .05 Alcohol volume in one beer= VBond = .6 ounces Ounce = 29.6 mL One "Beer" of alcohol = .6 ounces (Volume of alcohol in a 12 ounce beer) Pint = 16 ounces Wine or Scotch bottle = 25.4 ounces = 750 ml Pitcher = 64 ounces Gallon = 128 ounces Keg =1984 ounces = 15.5 gallonsConclusions can be read off from the table, such as the fact that the cheapest alcohol in terms of price per alcohol content is Charles Shaw wine.
In the following sections we show more examples of 2D tables.
 |
|---|
Caffeine Density Volume
mg mg/oz mg/oz
Coffee, brewed 163 20.4 8
Mtn. Dew Game Fuel 121 6.0 20
Red Bull 80 9.5 8.5
Espresso 77 51 1.5
Mountain Dew 54 4.5 12
Mello Yello 51 4.2 12
Tea (black) 42 5.2 8
Sunkist 41 3.4 12
Pepsi Cola 38 3.2 12
Arizona iced tea 38 1.9 20
Coca Cola 34 2.8 12
Coffee, decaf 6 .7 8
Sprite 0 0 12
 |
 |
 |
 |
|
|---|---|---|---|---|
A typical bottle of beer has a volume of 12 ounces, is 5% alcohol, and contains
.6 ounces of alcohol. We use this amount as a reference unit and define
.6 ounces of alcohol to be one "Bond".
Volume of the drink = V
Fraction of alcohol = F
Volume of alcohol = Valc = F V
Volume of one beer = Vbeer = 12 ounces
Alcohol fraction of beer = Fbeer = .05
Alcohol volume in one beer = VBond = .6 ounces
One "Bond" of alcohol = .6 ounces
One wine or Scotch bottle = 25.4 ounces = 750 ml
One ounce = 29.6 mL
Alcohol Volume Alcohol Alcohol $ $/Bond
fraction (oz) (oz) (Bonds)
Beer (12 oz) .05 12 .6 1 .67 .67 Budweiser
Wine glass .13 4.6 .6 1 8 8.0 Napa Valley
Scotch shot .40 1.5 .6 1 8 8.0 Laphroaig
Beer pitcher .05 64 3.2 5.3 16 3.0 Budweiser
Beer keg .05 1984 99.2 165.3 100 .60 Budweiser
Wine bottle .13 25.4 3.3 5.5 3 .55 Charles Shaw
Vodka bottle .40 25.4 10.1 16.9 15 .89 Smirnoff
Scotch bottle .40 25.4 10.1 16.9 50 3.0 Laphroaig
Distilled ethanol .95 25.4 24.1 40.2 15 .37 Everclear
Speed of light 2.9979e8 m/s
Gravitational constant 6.6738e-11 m3/kg/s2
Planck constant 6.6261e-34 J s
Earth surface gravity 9.8067 m/s
Electric force constant 8.9876e9 N m2 / C2
Magnetic constant 4 Pi e-7 N/A2
Proton mass 1.6726e-27 kg = 938.272 GeV
Neutron mass 1.6749e-27 kg = 939.565 GeV
Electron mass 9.1094e-31 kg
Electron charge 1.6022e-19 C
Atomic mass unit 1.6605e-27 kg
Bohr radius 5.2918e-11 m = hbar2 / (ElectronMass*ElectronCharge2*Ke)
Boltzmann constant 1.3806e-23 J/K
Avogadro number 6.0221e23 particles/mole
Gas constant 8.3145 J/K/mole
Stefan-Boltzmann constant 5.6704e-8 Watts/m2/K4
Wein constant 2.8978e-3 m K
Mole of Carbon-12 .012 kg Exact
Planck length 1.6162e-35 m
Planck mass 2.1765e-8 kg
Planck time 5.3911e-44 s
Planck charge 1.8755e-18 C
Planck temperature 1.4168e32 K
Water heat capacity 4200 J/kg/K
Steam heat capacity 2080 J/kg/K At 100 Celsius
Ice heat capacity 2110 J/kg/K At -10 Celsius
Air heat capacity 1004 J/kg/K
Stefan-Boltzmann 5.67e-8 Watts/meter2/Kelvin4
= (2π5/15) Boltzmann4 / SpeedOfLight2 / PlanckConstant3
Wein 2.898e-3 Kelvin meters
Electron spin 5.2729e-35 Joule seconds = PlanckConstant / (4 Pi)
Pi 3.14159
Euler number 2.71828
 |
|---|
For gases, the density at boiling point is used. Size data
 |
|---|
Copper atoms stack like cannonballs. We can calculate the atom size by assuming the atoms are shaped like either cubes or spheres. For copper atoms,
Density = D = 8900 kg/m3 Atomic mass unit= M0 = 1.661⋅10-27 kg Atomic mass = MA = 63.55 Atomic mass units Mass = M = MA⋅M0 = 9.785⋅10-26 kg Number density = N = D / M = 9.096⋅1028 atoms/m3 Cube volume = Υcube= 1 / N = 1.099⋅10-29 m3 Volume/atom if the atoms are cubes Cube length = L = Υ1/3cube = 2.22⋅10-10 m Side length of the cube Sphere fraction = f = π/(3√2) = .7405 Fraction of volume occupied by spheres in a stack o spheres Sphere volume = Υsph= Υcube f = 8.14⋅10-30 m3 = 4⁄3πR3 Volume/atom if the atoms are spheres Sphere radius = R = 1.25⋅10-10 m
 |
|---|
Modern mathematics and physics was launched when Simon Stevin popularized decimal numbers in Europe. Cartesian geometry and the calculus followed shortly after. Mathematics has been on a roll ever since.
Decimal numbers enable precise calculation, which is essential for science. Shortly after decimal numbers were popularized, the logarithm and the slide rule were invented. The slide rule enables fast multiplication and division.
 |
 |
 |
|
|---|---|---|---|
1585 Stevin popularizes decimal numbers in Europe
1614 Napier develops logarithm tables
1622 Oughtred develops the slide rule
1604 Galileo publishes the mathematical description of acceleration.
1637 Cartesian geometry published by Fermat and Descartes.
1684 Leibniz publishes the calculus
1687 Newton publishes the Principia Mathematica, which contained the calculus,
the laws of motion (F=MA), and a proof that planets orbit as ellipses.
 |
|---|
Energy/Mass Mass fraction
MJoule/kg
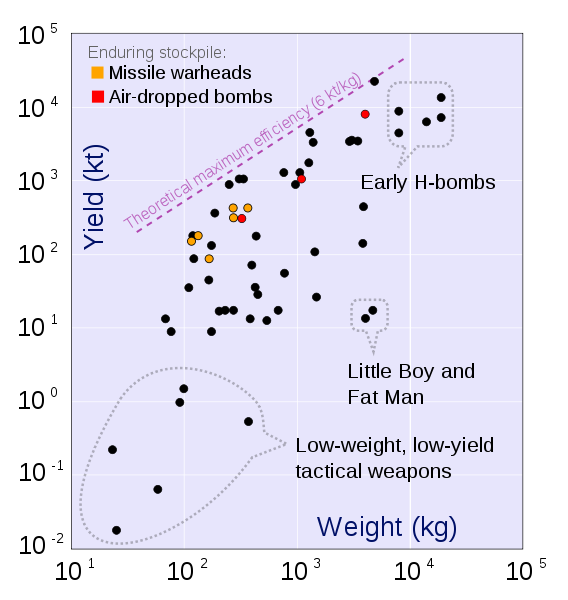
Antimatter 90,000,000,000 1
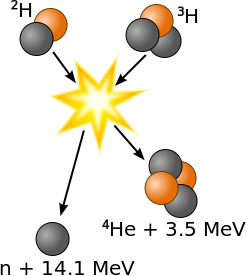
Fusion, D + Li6 268,000,000 .00298
Fusion bomb 25,000,000 .000278 Maximum practical yield of a bomb
Fission, U-235 83,000,000 .000918
Fission bomb 6,000,000 .000067 Maximum practical yield of a bomb
Fission, fast neutron 28,000,000 Fast neutrons, unenriched fuel
Fission, slow neutron 500,000 Slow neutrons, unenriched fuel
Nuclear battery, Co60 4,300,000 Half life 5.3 year
Nuclear battery, Pu238 2,260,000 Half life 88 year
Nuclear battery, Pu241 1,960,000 Half life 14.4 year
Nuclear battery, Sr90 590,000 Half life 29 year
Hydrogen 141.8
Methane 55.5 1 carbon. Natural gas
Ethane 51.9 2 carbons
Propane 50.4 3 carbons
Butane 49.5 4 carbons
Octane 47.8 8 carbons
Kerosene 46 12 carbons
Diesel 46 16 carbons
Oil 46 36 carbons
Fat 37 20 carbons. 9 Calories/gram
Pure carbon 32.8
Coal 32 Similar to pure carbon
Ethanol 29 7 Calories/gram
Wood 22
Sugar 17 4 Calories/gram
Protein 17 4 Calories/gram
Plastic explosive 8.0 HMX
Smokeless powder 5.2 Modern gunpowder
TNT 4.7
Black powder 2.6 Medieval gunpowder
Phosphocreatine .137 Recharges ATP
ATP .057 Adenosine triphosphate
Aluminum capacitor .010
Spring .0003
Battery, aluminum-air 4.68
Battery, Li-S 1.44
Battery, Li-ion .8
Battery, Li-polymer .6
Battery, Alkaline .4
Battery, Lead acid .15
"Mass fraction" is the fraction of mass converted to energy, by E=MC2.
 |
|---|
Equations can often be derived using units. For example, what is the formula for kinetic energy? The variables that will be present in the formula are:
Mass = M (kg) Velocity = V (meters/second) Kinetic energy = E (Joules = kg meters2/second2) Dimensionless constant = K (Unitless)Assume the formula as the form
E = K Mm VvFor some value of m and v. The values that gives units of energy are:
E = K M1 V2Units arguments often give the right formula up to a dimensionless constant and a more involved derivation is usually required to produce the constant. The formula with the dimensionless constant included can always be found on Wikipedia. For the kinetic energy, K=½ and E = ½ M V2.
Another example of using units to derive formulae is the aerodynamic drag force. The variables that will be present in the formula are:
Velocity = V meters/second Cross sectional area = A meters2 Density of air = D = 1.22 kg/meter3 Drag force = F Newtons = kg meters/second2 Dimensionless constant= K UnitlessAssume the formula has the form
F = K Dd Aa Vvfor some value of {d,a,v}. The values that give units of force are
F = K D1 A1 V2
Aerodynamic drag force = 1/2 Density CrossSection Velocity2 Aerodynamic drag power = 1/2 Density CrossSection Velocity3 Gravitational force = -G Mass1 Mass2 / Distance2 Gravitational energy = -G Mass1 Mass2 / Distance Gravitational self-energy = 3/5 G Mass2 / Radius For a sphere of uniform density Kinetic energy = 1/2 Mass Velocity2 Sound Speed = [Γ Pressure / Density]1/2 Γ=7/5 for air Wave speed for a string = [Tension Length / Mass]1/2
 |
 |
|---|---|
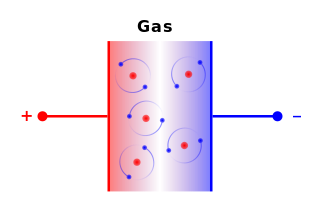
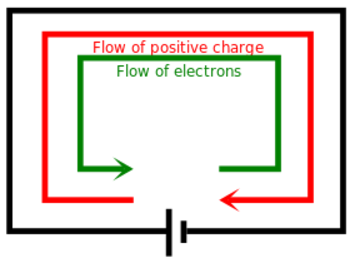
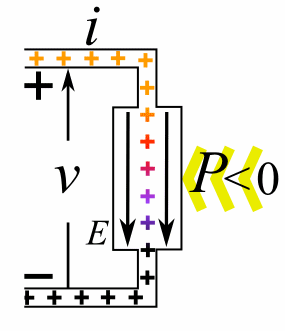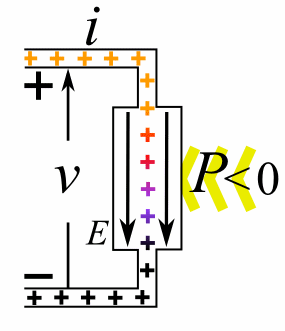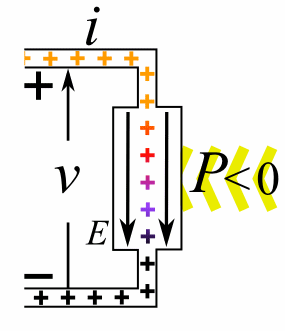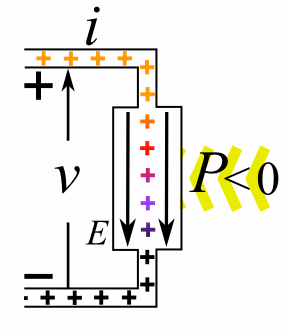
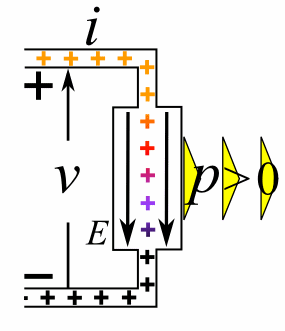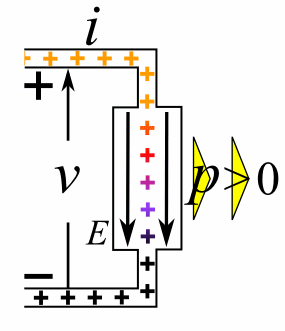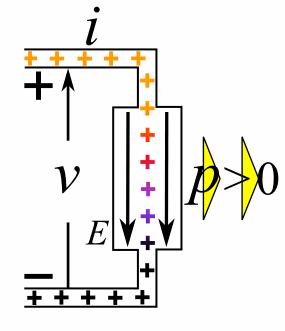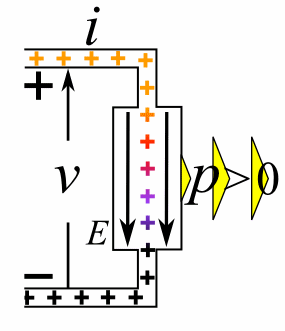
A moving charge is an "electric current". In an electric circuit, a battery moves electrons through a wire.
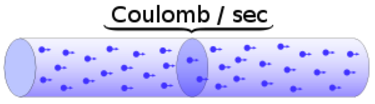
Charge = Q Time = T Electric current = I = Q / T (Coulombs/second)The current from a positive charge moving to the right is equivalent to that from a negative charge moving to the left.
 |
 |
|---|---|
Charge = Q Coulombs
Voltage = V Volts
Energy = E = VQ Joules
Time = T seconds
Current = I = Q/T Amperes
Resistance = R = V/I Ohms
Power = P = QV/T Watts
= IV
= V2/R
= I2R
Ohm's Law: V = IR
 |
 |
|---|---|
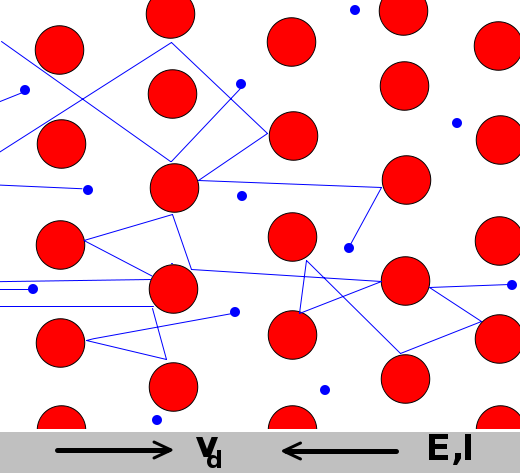
In a superconductor, electrons move without interference.
In a resistor, electrons collide with atoms and lose energy.
Resistance (Ohms)
Copper wire .02 1 meter long and 1 mm in diameter
1 km power line .03
AA battery .1 Internal resistance
Light bulb 200
Human 10000
 |
|---|