




 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
 |
 |
|
|---|---|---|---|
MJoules Rocket Shock Density Boil
/kg km/s km/s g/cm3 Kelvin
Beryllium+ O2 23.2 5.3
Aluminum + O2 15.5
Magnesium+ O2 14.8
Hydrogen + O2 13.2 4.56 .07 20
Kerosene + O3 12.9
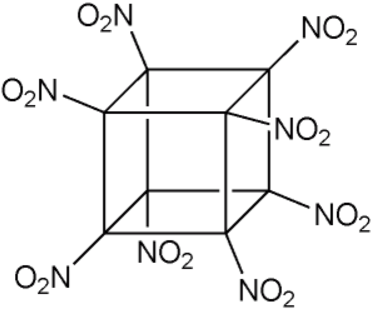
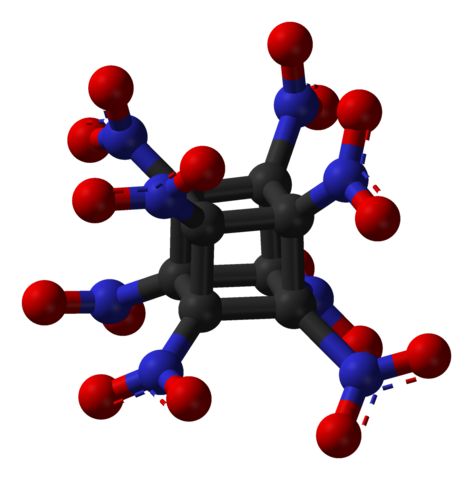
Octanitrocubane 11.2 10.6 1.95
Methane + O2 11.1 3.80 .42 112 CH4
Octane + O2 10.4 .70 399 C8H18
Kerosene + O2 10.3 3.52 .80 410 C12H26
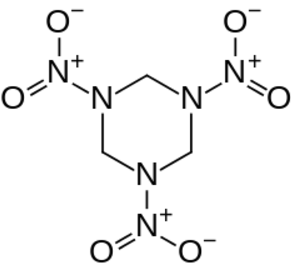
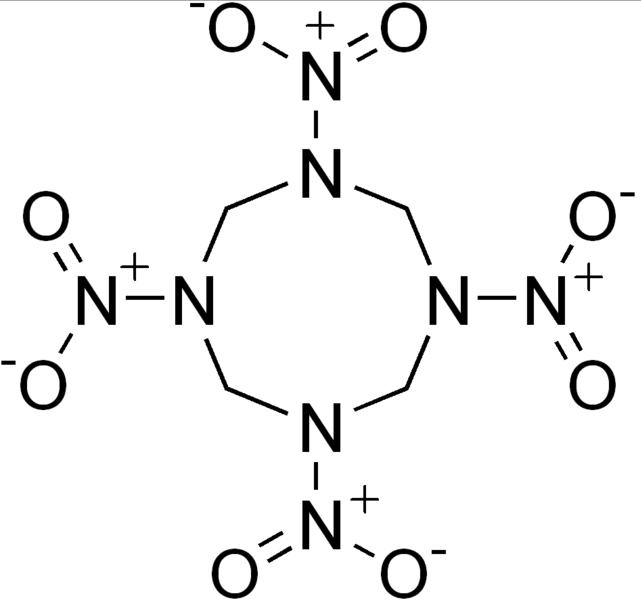
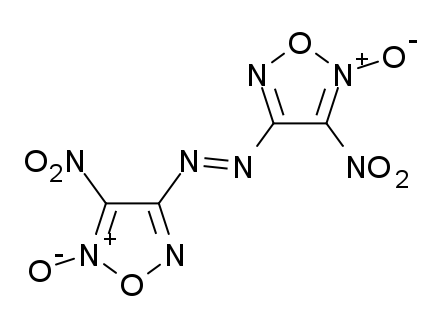
Dinitrodiazeno. 9.2 10.0 1.98
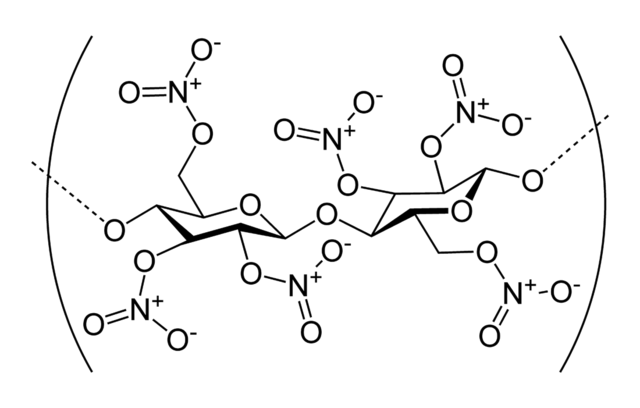
C6H6N12O12 9.1 1.96 China Lake compound
Kerosene + H2O2 8.1 3.2
Kerosene + N2O4 8.0 2.62
HMX (Octogen) 8.0 3.05 9.1 1.86
RDX (Hexagen) 7.5 2.5 8.7 1.78
Al + NH4NO3 6.9
Nitroglycerine 7.2 8.1 1.59 Unstable
PLX 6.5 1.14 95% CH3NO2 + 5% C2H4(NH2)2
Composition 4 6.3 8.04 1.59 91% RDX. "Plastic explosive"
Kerosene + N2O 6.18
Dynamite 5.9 7.2 1.48 75% Nitroglycerine + stabilizer
PETN 5.8 8.35 1.77
Smokeless powder 5.2 6.4 1.4 Used after 1884. Nitrocellulose
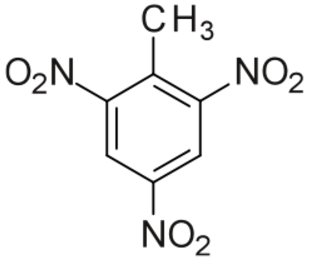
TNT 4.7 6.9 1.65 Trinitrotoluene
Al + Fe2O3 4.0 Thermite
H2O2 2.7 1.59 1.45 423 Hydrogen peroxide
Black powder 2.6 .6 1.65 Used before 1884
Al + NH4ClO4 2.6
NH4ClO4 2.5
N2O 1.86 1.76
N2H4 1.6 2.2 1.02 387 Hydrazine
Bombardier beetle .4 Hydroquinone + H2O2 + protein catalyst
N2O4 .10 1.45 294
Rocket: Rocket exhaust speed
Shock: Shock speed
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|
 |
 |
|---|---|
High explosives have a large shock velocity.
MJoules Shock Density
/kg km/s g/cm3
Octanitrocubane 11.2 10.6 1.95
Dinitrodiazeno. 9.2 10.0 1.98
C6H6N12O12 9.1 1.96 China Lake compound
HMX (Octogen) 8.0 9.1 1.86
RDX (Hexagen) 7.5 8.7 1.78
PLX 6.5 1.14 95% CH3NO2 + 5% C2H4(NH2)2
Composition 4 6.3 8.04 1.59 91% RDX. "Plastic explosive"
Dynamite 5.9 7.2 1.48 75% Nitroglycerine + stabilizer
PETN 5.8 8.35 1.77
The best oxidizer is liquid oxygen, and the exhaust speed for various fuels when burned with oxygen is:
Exhaust Energy Density of fuel + oxidizer
speed /mass
km/s MJ/kg g/cm3
Hydrogen H2 4.46 13.2 .32
Methane CH4 3.80 11.1 .83
Ethane C2H6 3.58 10.5 .9
Kerosene C12H26 3.52 10.3 1.03
Hydrazine N2H4 3.46 1.07
Liquid hydrogen is usually not used for the ground stage of rockets because of
its low density.
We use kerosene as a standard fuel and show the rocket speed for various oxidizers. Some of the oxidizers can be used by themselves as monopropellants.
Energy/Mass Energy/Mass Rocket Rocket Boil Density
with kerosene as monopropellant with kerosene as monopropellant Kelvin g/cm3
MJoule/kg MJoule/kg km/s km/s
O3 12.9 2.97 161
O2 10.3 0 3.52 0 110 1.14
H2O2 8.1 2.7 3.2 1.6 423 1.45
N2O4 8.00 .10 2.62 294 1.44
N2O 6.18 1.86 1.76 185
N2H4 - 1.58 2.2 387 1.02
MJoules Rocket Density
/kg km/s g/cm3
C6H6N12O12 9.1 1.96 China Lake compound
HMX (Octogen) 8.0 3.05 1.86
RDX (Hexagen) 7.5 2.5 1.78
Al + NH4ClO4 2.6
NH4ClO4 2.5
NH3OHNO3 2.5 1.84 Hydrxyammonium nitrate
Al + NH4NO3 6.9
NH4NO3 1.4 2.0 1.12 Ammonium nitrate
~808 Qing Xuzi publishes a formula resembling gunpower, consisting of
6 parts sulfur, 6 parts saltpeter, and 1 part birthwort herb (for carbon).
~850 Incendiary property of gunpower discovered
1132 "Fire lances" used in the siege of De'an, China
1220 al-Rammah of Syria publishes "Military Horsemanship and Ingenious War
Devices", describes the purification of potassium nitrate by
adding potassium carbonate with boiling water, to precipitate out
magnesium carbonate and calcium carbonate.
1241 Mongols use firearms at the Battle of Mohi, Hungary
1338 Battle of Arnemuiden. First naval battle involving cannons.
1346 Cannons used in the Siege of Calais and the Battle of Crecy
1540 Biringuccio publishes "De la pirotechnia", giving recipes for gunpowder
1610 First flintlock rifle
1661 Boyle publishes "The Sceptical Chymist", a treatise on the
distinction between chemistry and alchemy. It contains some of the
earliest modern ideas of atoms, molecules, and chemical reaction,
and marks the beginning of the history of modern chemistry.
1669 Phosphorus discovered
1774 Lavoisier appointed to develop the French gunpowder program. By 1788
French gunpowder was the best in the world.
1832 Braconnot synthesizes the first nitrocellulose (guncotton)
1846 Nitrocellulose published
1847 Sobrero discovers nitroglycerine
1862 LeConte publishes simple recipes for producing potassium nitrate.
1865 Abel develops a safe synthesis of nitrocellulose
1867 Nobel develops dynamite, the first explosive more powerful than black powder
It uses diatomaceous earth to stabilize nitroglycerine
1884 Vieille invents smokeless gunpowder (nitrocellulose), which is 3 times
more powerful than black powder and less of a nuisance on the battlefield.
1902 TNT first used in the military. TNT is much safer than dynamite
1930 RDX appears in military applications
1942 Napalm developed
1949 Discovery that HMX can be synthesized from RDX
1956 C-4 explosive developed (based on RDX)
1999 Eaton and Zhang synthesize octanitrocubane and heptanitrocubane
Black powder = .75 KNO3 + .19 Carbon + .06 Sulfur
Above 550 Celsius, potassium nitrate decomposes. 2 KNO3 ↔ 2 KNO2 + O2.
 |
 |
 |
|
|---|---|---|---|
 |
 |
 |
 |
|---|---|---|---|
Potassium nitrate KNO3 75% (Saltpeter) Charcoal C7H4O 15% Sulfur S 10% Oversimplified equation: 2 KNO3 + 3 C + S → K2S + N2 + 3 CO2 Realistic equation: 6 KNO3 + C7H4O + 2 S → KCO3 + K2SO4 + K2S + 4 CO2 + 2 CO + 2 H2O + 3 N2Nitrite (NO3) is the oxidizer and sulfur lowers the ignition temperature.
MJoules
/kg
Hydrogen + Oxygen 13.16
Gasoline + Oxygen 10.4
Mass Energy Energy/Mass
kg MJ MJ/kg
MOAB 9800 46000 4.7 8500 kg of fuel
 |
 |
 |
|---|---|---|
 |
 |
 |
|---|---|---|
Form Ignition Density
(Celsius)
White 30 1.83
Red 240 1.88
Violet 300 2.36
Black 2.69
Red phosphorus is formed by heating white phosphorus to 250 Celsius or by
exposing it to sunlight. Violet phosphorus is formed by heating red phosphorus
to 550 Celsius. Black phosphorus is formed by heating white phosphorus at a
pressure of 12000 atmospheres. Black phosphorus is least reactive form and it
is stable below 550 Celsius.
 |
 |
 |
 |
 |
|
|---|---|---|---|---|---|
The safety match was invented in 1844 by Pasch. The match head cannot ignite by itself. Ignitition is achieved by striking it on a rough surface that contains red phosphorus. When the match is struck, potassium chlorate in the match head mixes with red phosphorus in the abrasive to produce a mixture that is easily ignited by friction. Antimony trisulfide is added to increase the burn rate.
Match head Fraction Striking surface Fraction Potassium chlorate KClO3 .50 Red phosphorus .5 Silicon filler Si .4 Abrasive .25 Sulfur S small Binder .16 Antimony3 trisulfide Sb2S3 small Neutralizer .05 Neutralizer small Carbon .04 Glue smallA "strike anywhere" match has phosphorus in the match head in the form of phosphorus sesquisulfide (P4S3) and doesn't need red phosphorus in the striking surface. P4S3 has an ignition temperature of 100 Celsius.
 |
 |
.jpg) |
|---|---|---|
Before the invention of iron, fires were started by striking flint (quartz) with pyrite to generate sparks. Flintlock rifles work by striking flint with iron. With the discovery of cerium, ferrocerium replaced iron and modern butane lighters use ferrocerium, which is still referred to as "flint".
Cerium .38 Ignition temperature of 165 Celsius Lanthanum .22 Iron .19 Neodymium2 .04 Praseodymium .04 Magnesium .04
 |
|---|
Nitrous oxide is stored as a cryogenic liquid and injected along with gaoline into the combustion chamber. Upon heating to 300 Celsius the nitrous oxide decomposes into nitrogen and oxygen gas and releases energy. The oxygen fraction in this gas is higher than that in air (1/3 vs. .21) and the higher faction allows for more fuel to be consumed per cylinder firing.
Air density = .00122 g/cm3 Nitrous oxide gas density = .00198 g/cm3 Diesel density = .832 g/cm3 Gasoline density = .745 g/cm3 Diesel energy/mass = 43.1 MJoules/kg Gasoline energy/mass = 43.2 MJoules/kg Nitrous oxide boiling point = -88.5 Celsius Air oxygen fraction = .21 Nitrous oxide oxygen fraction= .33 Nitrous oxide decompose temp = 300 Celsius Nitrous oxide liquid pressure= 52.4 Bars Pressure required to liquefy N2O at room temperature
 |
 |
 |
|---|---|---|
Hydroquinone and peroxide are stored in 2 separate compartments are pumped into the reaction chamber where they explode with the help of protein catalysts. The explosion vaporizes 1/5 of the liquid and expels the rest as a boiling drop of water, and the p-quinone in the liquid damages the foe's eyes. The energy of expulsion pumps new material into the reaction chamber and the process repeats at a rate of 500 pulses per second and a total of 70 pulses. The beetle has enough ammunition for 20 barrages.
2 H2O2 → 2 H2O + O2 (with protein catalyst) C6H4(OH)2 → C6H4O2 + H2 (with protein catalyst) O2 + 2 H2 → 2 H2O Firing rate = 500 pulses/second Number of pulses in one barrage = 70 Firing time = .14 seconds Number of barrages = 20
 |
 |
 |
 |
|
|---|---|---|---|---|
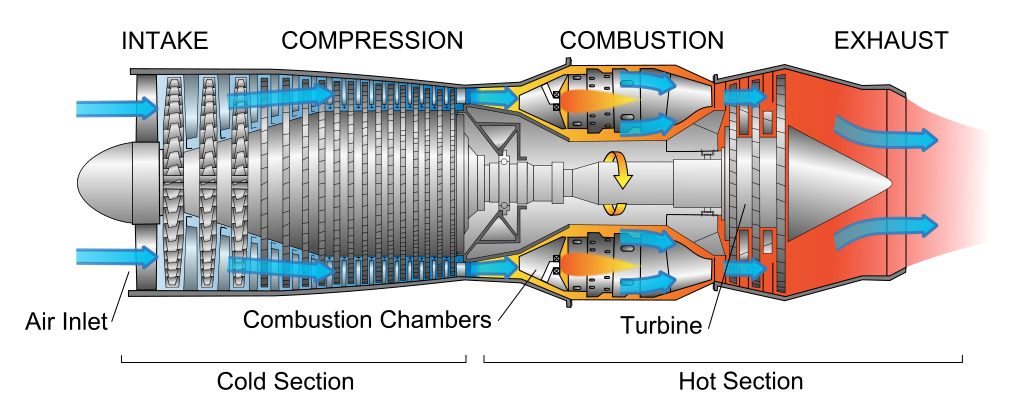
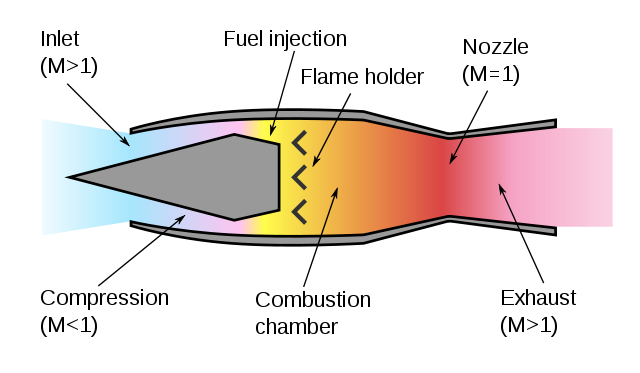
A turbojet engine compresses air before burning it to increase the flame speed and make it burn explosively. A ramjet engine moving supersonically doesn't need a turbine to achieve compression.
 |
 |
|---|---|
Airbus A350 compression ratio = 52 Air density at sea level = 1 bar Air density at 15 km altitude = .25 bar Air density in A350 engine = 13 barFrom the thermal flame theory of Mallard and Le Chatelier,
Temperature of burnt material = Tb Temperature of unburnt material = Tu Temperature of ignition = Ti Fuel density = Dfuel Oxygen density = Doxygen Reaction coefficient = C Reaction rate = R = C Dfuel Doxygen Thermal diffusivity = Q = 1.9⋅10-5 m2/s Flame speed = V V2 = Q C Dfuel Doxygen (Tb - Ti) / (Ti - Tu)
 |
 |
 |
 |
 |
|---|---|---|---|---|
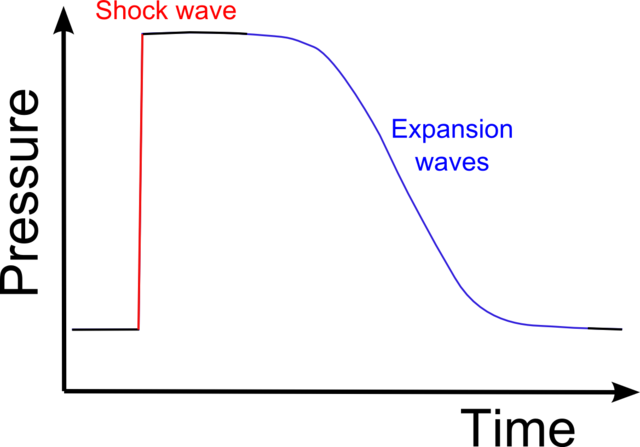
If the pressure front moves supersonically then the front forms a discontinuous shock, where the pressure makes a sudden jump as the shock passes.
Metal powder is often included with explosives.
Energy/mass Energy/mass
not including including
oxygen oxygen
(MJoule/kg) (MJoule/kg)
Hydrogen 113.4 12.7
Gasoline 46.0 10.2
Beryllium 64.3 23.2
Aluminum 29.3 15.5
Magnesium 24.5 14.8
Carbon 12.0 3.3
Lithium 6.9 3.2
Iron 6.6 4.6
Copper 2.0 1.6
 |
 |
 |
 |
 |
|---|---|---|---|---|
 |
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|---|
 |
 |
 |
 |
 |
 |
 |
|---|---|---|---|---|---|---|
 |
 |
 |
 |
 |
|---|---|---|---|---|
 |
|---|
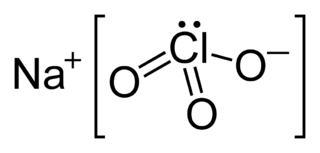
An oxygen candle is a mixture of sodium chlorate and iron powder, which when ignited smolders at 600 Celsius and produces oxygen at a rate of 6.5 man-hours of oxygen per kilogram of mixture. Thermal decomposition releases the oxygen and the burning iron provides the heat. The products of the reaction are NaCl and iron oxide.
 |
|---|