
 |
|---|
 |
|---|
Charge
Proton +1
Neutron 0
Electron -1
Antineutrino 0
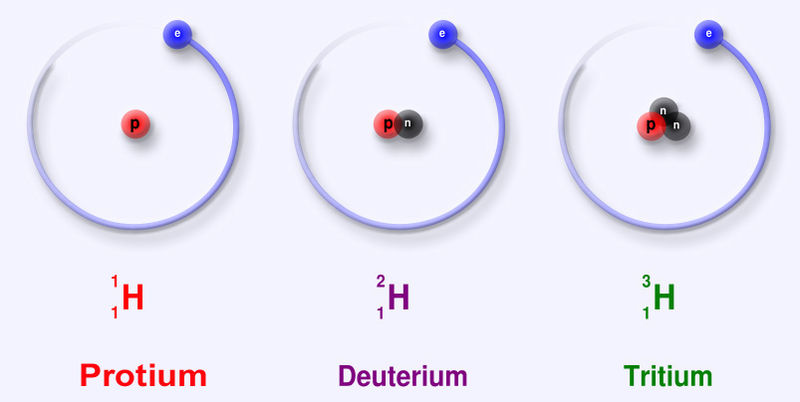
Isotopes:
Symbol Protons Neutrons Half life
Electron e 0 0 stable
Neutron N 0 1 886 seconds
Proton P 1 0 stable
Deuterium D 1 1 stable
Tritium T 1 2 12.3 years
Helium-3 He3 2 1 stable
Helium-4 He4 2 2 stable
Lithium-6 Li6 3 3 stable
Lithium-7 Li7 3 4 stable
Carbon-12 C12 6 6 stable
Oxygen-16 O16 8 8 stable
 |
|---|
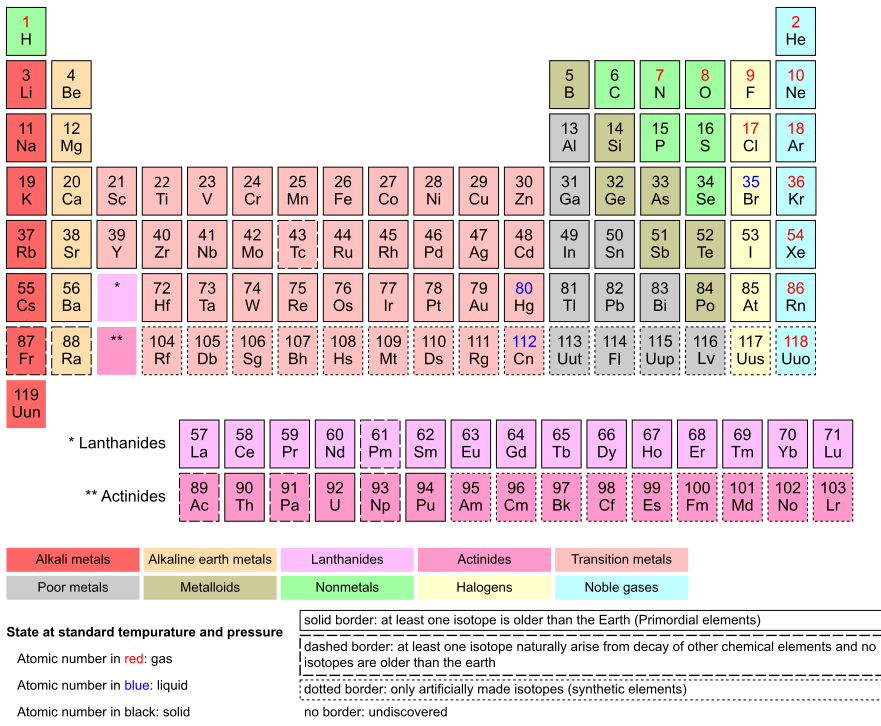
Each number corresponds to the number of protons.
Teaching simulation for isotopes at phet.colorado.edu
 |
|---|
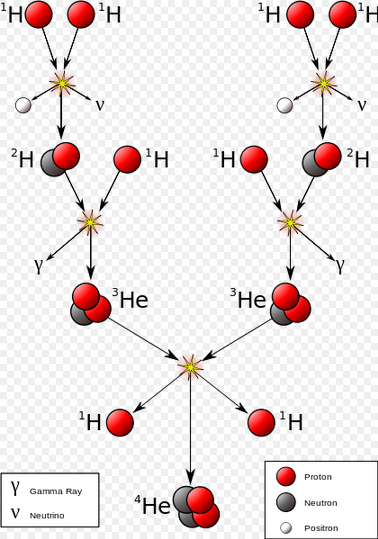
Hydrogen fusion requires a temperature of at least 4 million Kelvin, which requires an object with at least 0.08 solar masses. This is the minimum mass to be a star.
P + P --> D + Positron + Neutrino + .42 MeV P + D --> He3 + Photon + 5.49 MeV He3 + He3 --> He4 + P + P + 12.86 MeV 1 MeV = 106 eV = 1.602*10-13 Joules
 |
|---|
As the core of a star star runs out of hydrogen it contracts and heats, and helium fusion begins when the temperature reaches 10 million Kelvin
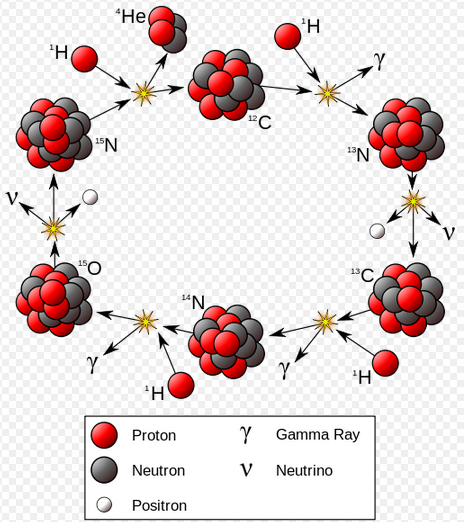
At temperatures above 17 million Kelvin, carbon-catalyzed fusion happens faster than proton-proton fusion. This occurs in stars more massive than 1.3 solar masses.
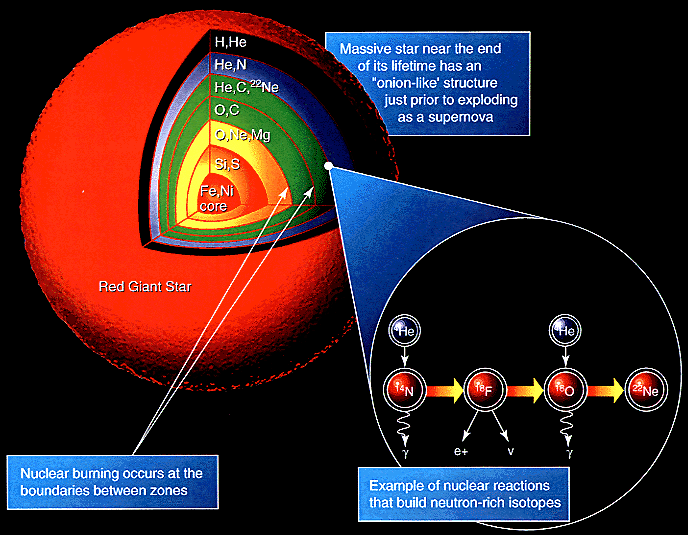 |
|---|
A heavy star continues to fuse elements until it reaches Iron-56. Beyond this, fusion absorbs energy rather than releasing it, triggering a runaway core collapse that fuses elements up to Uranium. If the star explodes as a supernova then these elements are ejected into interstellar space.
Star type Mass Luminosity Color Temp Lifetime Death Remnant Size of Output
(solar (solar (Kelvin) (billions remnant
masses) luminosities) of years)
Brown Dwarf <0.08 1000 immortal
Red Dwarf 0.1 .0001 red 2000 1000 red giant white dwarf Earth-size
The Sun 1 1 white 5500 10 red giant white dwarf Earth-size light elements
Blue star 10 10000 blue 10000 0.01 supernova neutron star Manhattan heavy elements
Blue giant 20 100000 blue 20000 0.01 supernova black hole Central Park heavy elements
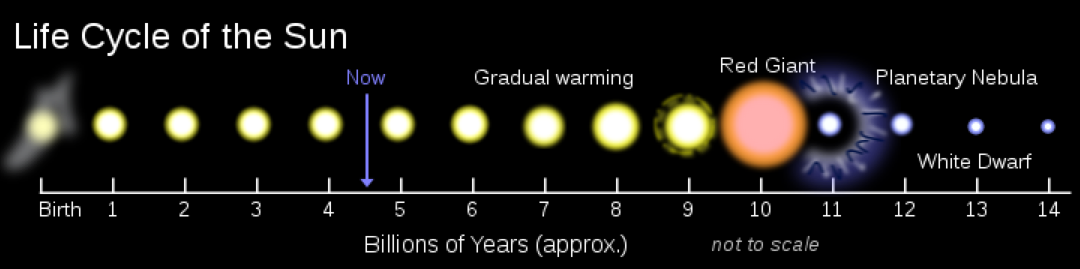
Fate of stars, with mass in solar masses:
Mass < 9 --> End as red giants and then turn white dwarf.
9 < Mass --> End as supernova
9 < Mass < 20 --> Remnant is a neutron star.
20 < Mass --> Remnant is a black hole.
130 < Mass < 250 --> Pair-instability supernova (if the star has low metallicity)
250 < Mass --> Photodisintegration supernova, producing a black hole and relativistic jets.
 |
 |
|---|---|
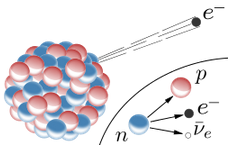
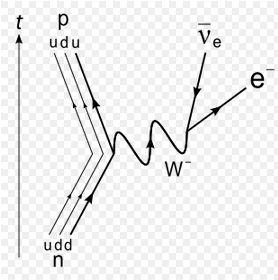
The weak force can convert a neutron into a proton.
From the point of view of nucleons: Neutron --> Proton + electron + antineutrino From the point of view of quarks: Down quark --> Up quark + electron + antineutrino
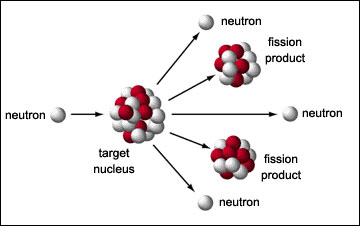
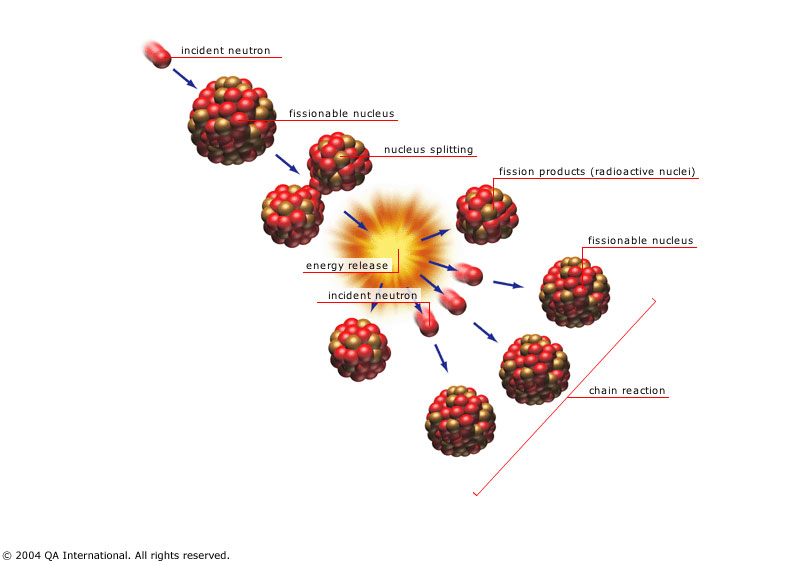
A neutron triggers the fission of Uranium-235 and plutonium-239, releasing energy and more neutrons.
 |
 |
|---|---|
 |
 |
|---|---|
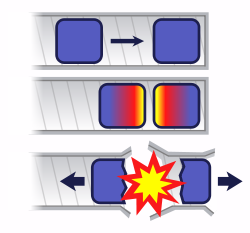
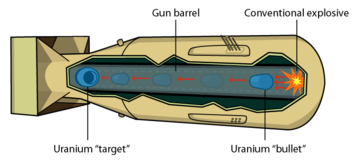
Two pieces of uranium, each with less than a critical mass, are brought together in a cannon barrel.
If the uranium is brought together too slowly, the bomb fizzles.
 |
 |
|---|---|
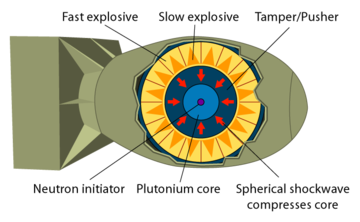
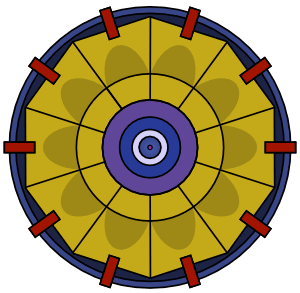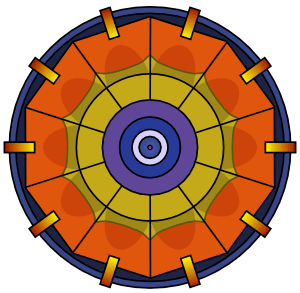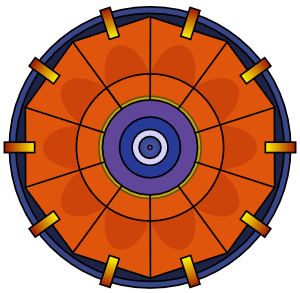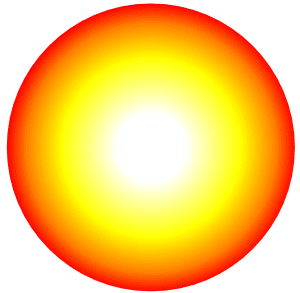
Plutonium is more difficult to detonate than uranium. Plutonium detonation requires a spherical implosion.
 |
|---|
Blue elements are unstable with a half life much less than the age of the solar system.
The only elements heavier than Bismuth that can be found on the Earth are Thorium and Uranium, and these are the only elements that can be tapped for fission energy.
Natural Thorium is 100% Thorium-232
Natural Uranium is .72% Uranium-235 and 99.3% Uranium-238.
Plutonium doesn't exist in nature.
Protons Neutrons Halflife Critical Isotope
(10^6 yr) mass (kg) fraction
Thorium-232 90 142 14000 - 1.00 Absorbs neutron -> U-233
Uranium-233 92 141 .160 16 - Fission chain reaction
Uranium-235 92 143 700 52 .0072 Fission chain reaction
Uranium-238 92 146 4500 - .9927 Absorbs neutron -> Pu-239
Plutonium-238 94 144 .000088 - - Produces power from radioactive heat
Plutonium-239 94 145 .020 10 - Fission chain reaction
The elements that can be used for fission energy are the ones with a critical
mass. These are Uranium-233, Uranium-235, and Plutonium-239.
Uranium-233 and Plutonium-239 can be created in a breeder reactor.
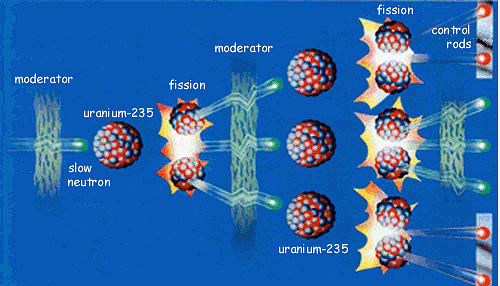
Thorium-232 + Neutron -> Uranium-233 Uranium-238 + Neutron -> Plutonium-239The "Fission" simulation at phet.colorado.edu illustrates the concept of a chain reaction.
Natural uranium is composed of .7% Uranium-235 and the rest is Uranium-238. Uranium-235 can be separated from U-238 using centrifuges, calutrons, or gas diffusion chambers. Uranium-235 is easy to detonate. A cannon and gunpowder gets it done.
Plutonium-239 is difficult to detonate, requiring a perfect spherical implosion. This technology is beyond the reach of most rogue states.
Uranium-233 cannot be used for a bomb and is hence not a proliferation risk.
Plutonium-238 emits alpha particles, which can power a radioisotope thermoelectric generator (RTG). RTGs based on Plutonium-238 generate 540 Watts/kg and are used to power spacecraft.
Creating Plutonium-239 and Uranium-233:
Uranium-238 + Neutron -> Plutonium-239 Thorium-232 + Neutron -> Uranium-233 Detail: Uranium-238 + Neutron -> Uranium-239 Uranium-239 -> Neptunium-239 + Electron + Antineutrino Halflife = 23 mins Neptunium-239 -> Plutonium-239 + Electron + Antineutrino Halflife = 2.4 days Thorium-232 + Neutron -> Thorium-233 Thorium-233 -> Protactinium-233 + Electron + Antineutrino Halflife = 22 mins Protactinium-233 -> Uranium-233 + Electron + Antineutrino Halflife =
1885 Rontgen discovers X-rays
1899 Rutherford discovers alpha and beta rays
1903 Rutherford discovers gamma rays
1905 E=mc^2. Matter is equivalent to energy
1909 Nucleus discovered by the Rutherford scattering experiment
1932 Neutron discovered
1933 Nuclear fission chain reaction envisioned by Szilard
1934 Fermi bombards uranium with neutrons and creates Plutonium. First
successful example of alchemy
1938 Fission discovered by Hahn and Meitner
1938 Bohr delivers news of fission to Princeton and Columbia
1939 Fermi constructs the first nuclear reactor in the basement of Columbia
1939 Szilard and Einstein write a letter to President Roosevelt advising
him to consider nuclear fission
1942 Manhattan project started
1942-1945 German nuclear bomb project goes nowhere
1945 Two nuclear devices deployed by the United States
1 ton of TNT 4*10^9 Joules 1 ton of gasoline 4*10^10 Joules North Korea fission device 0.5 kilotons TNT 10 kg uranium fission bomb 10 kilotons TNT 10 kg hydrogen fusion bomb 10 megatons TNT Tunguska asteroid strike 15 megatons TNT 50 meter asteroid Chixulub dinosaur extinction 100 trillion tons TNT 10 km asteroid
A tokamak fusion reactor uses magnetic fields to confine a hot plasma so that fusion can occur in the plasma.
 |
 |
|---|---|
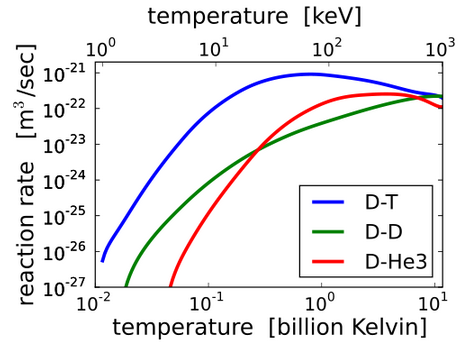
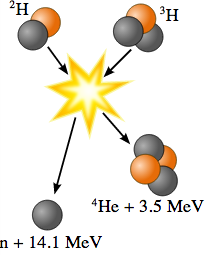
The fusion reaction that occurs at the lowest temperature and has the highest reaction rate is
Deuterium + Tritium --> Helium-4 + Neutron + 17.590 MeVbut the neutrons it produces are a nuisance to the reactor.
A potential fix is to have "liquid walls" absorb the neutrons (imagine a waterfall of neutron-absorbing liquid lithium cascading down the walls of the reactor).
The fusion reactions that don't produce neutrons are
Energy Coulomb
yield energy
(MeV)
P + P -> D + Positron .42 1 Slow because requires the weak force
P + D -> He3 + Photon 5.49 1 Slow because requires the electromagnetic force
D + He3 -> He4 + P 18.353 2 D+D side reactions produce neutrons
P + Li6 -> He4 + He3 4.0 3
P + Li7 -> He4 + He4 17.2 3
D + Li6 -> He4 + He4 22.4 3 D+D side reactions produce neutrons
He3 + He3 -> He4 + P + P 12.860 4 He3 is rare
P + B11 -> He4 + He4 + He4 8.7 5
He3 + Li6 -> He4 + He4 + P 16.9 6
P + N15 -> C12 + He4 5.0 7
"Coulomb energy" is the product of the charges of the two reactants, in units
of proton charge. The lower the energy, the easier it is to fuse the nuclei.
This can be seen with
the Rutherford
scattering simulation.Helium-3 is rare on the Earth and abundant on the moon.
A nuclear fusion bomb contains deuterium and lithium-6 and the reaction is catalyzed by a neutron.
N + Li6 -> He4 + T + 4.87 MeV T + D -> He4 + N + 17.56 MeV Total energy released = 22.43 MeV Nucleons = 8 Energy / Nucleon = 22.434 / 8 = 2.80
A rocket ideally produces as much energy per mass as possible, which is reflected in the fusion "energy per nucleon". The reactions that have the best energy per nucleon are
Energy Energy per
yield nucleon
(MeV) (MeV)
D + He3 -> He4 + P 18.353 3.67
D + T -> He4 + N 17.590 3.52
D + Li6 -> He4 + He4 22.4 2.80
T + He3 -> He4 + D 14.320 2.39 41%
P + Li7 -> He4 + He4 17.2 2.15
He3 + He3 -> He4 + P + P 12.860 2.14
T + He3 -> He4 + P + N 12.096 2.02 59%
The best choice is D + He3 and the next best choice is D + T.
Energy Energy per
yield nucleon
(MeV) (MeV)
P + P -> D + Positron .42 .21
P + D -> He3 + Photon 5.49 1.83
P + T -> He3 + N -.764
P + Li6 -> He4 + He3 4.0 .57
P + Li7 -> He4 + He4 17.2 2.15
P + B11 -> He4 + He4 + He4 8.7 .72
D + D -> T + P 4.033 1.01 50%
-> He3 + N 3.269 .81 50%
D + T -> He4 + N 17.590 3.52
D + He3 -> He4 + P 18.353 3.67
D + Li6 -> He4 + He4 22.4 2.80
T + T -> He4 + N + N 11.332 1.89
T + He3 -> He4 + P + N 12.096 2.02 59%
-> He4 + D 14.320 2.39 41%
He3 + He3 -> He4 + P + P 12.860 2.14
He3 + Li6 -> He4 + He4 + P 16.9 1.88
N + Li6 -> T + He4 4.784 .68
N + Li7 -> T + He4 + N -2.467
Mass of nucleus Mass of atom Half life Binding energy
(AMU) (AMU) per nucleon (MeV)
Electron .00054858
Neutron 1.00866492 886 seconds 0
Proton 1.00727647 0
Hydrogen 1.00727647 1.00782504 0
Deuterium 2.01355321 2.01410178 1.11226
Tritium 3.01550071 3.01604928 12.3 years 2.82727
Helium-3 3.01493173 3.0160293 2.57269
Helium-4 4.00150485 4.002602 7.07392
Lithium-6 6.01347537 6.01512280 5.33257
Lithium-7 7.01435712 7.01600455 5.60637
Beryllium-8 7*10^−17 s 7.06244
Beryllium-9 6.46278
Boron-10 6.47508
Boron-11 6.92771
Carbon-12 7.68015
Carbon-13 7.46986
Carbon-14 5730 years 7.52033
Oxygen-16 7.97622
Oxygen-17 7.75075
Oxygen-18 7.76707
Iron-56 8.79
Uranium-235 7.59
Uranium-238 7.57
1 MeV = 10^6 eV = 1.602*10^-13 Joules
If no half-life is given, the nucleus is stable
1 atomic mass unit (AMU) = 1.660538921*10^-27 kg = 931.494061 MeV
Parts Halflife Decay Neutron Result of Halflife
per (thousand energy absorb neutron (thousand
thousand years) (MeV) (barns) absorption years)
Caesium-135 69 1500 .27 8.3 Barium-136
Caesium-137 63 .030 1.2 .11 Barium-138
Technetium-99 61 210 .29 20 Ruthenium-100 stable
Zirconium-93 55 1500 .091 2.7 Niobium-94 20.3
Strontium-90 45 .029 2.8 .90 Zirconium-91 stable
Palladium-107 12.5 6500 .033 1.8 Silver-108 .418
Iodine-129 8.4 15700 .194 18 Xenon-130 ?
Samarium-151 5.3 .097 .077 15200 Europium-152 .0135
Krypton-85 2.2 .011 .69 1.7
Tin-126 1.1 230 4.0 < .1
Selenium-79 .447 330 .15 < .1
Europium-155 .80 .0048 .25 3950
Cadmium-113 .008 .014 .32 20600
Tin-121 .0005 .044 .39 ?
"Neutron absorption" is the cross section for a nucleus to capture a thermal
neutron.
All of the radioactive fission products decay by beta decay.
If the neutron cross section is 8 barnes or higher then the nucleus can potentially be transmuted into a nonradioactive nucleus.
Strontium-90 is ideal for Radioisotope Thermoelectric Generators (RTGs). www.jaymaron.com/rockets/rockets.html
The most troublesome fission products are the ones that can't be transmuted. Chief among these are Caesium-137, Zirconium-93, Niobium-94, Strontium-90, Zirconium-91, and Palladium-107.
Fission Fusion
U.S.A. 1945 1954
Germany Attempted fission in 1944 & failed
Russia 1949 1953
Britain 1952 1957
France 1960 1968
China 1964 1967
India 1974 Uranium
Israel 1979 ? Undeclared. Has both fission and fusion weapons
South Africa 1980 Dismantled in 1991
Iran 1981 Osirak reactor to create Plutonium. Reactor destroyed by Israel
Pakistan 1990 Centrifuge enrichment of Uranium. Tested in 1998
Built centrifuges from stolen designs
Iraq 1993 Magnetic enrichment of Uranium. Dismantled after Gulf War 1
Iraq 2003 Alleged by the United States. Proved to be untrue.
North Korea 2006 Obtained plutonium from a nuclear reactor. Detonation test fizzled
Also acquired centrifuges from Pakistan
Also attempting to purify Uranium with centrifuges
Syria 2007 Nuclear reactor destroyed by Israel
Iran 2009? Attempting centrifuge enrichment of Uranium.
Libya -- Attempted centrifuge enrichment of Uranium. Dismantled before completion.
Cooperated in the investigation that identified
Pakistan as the proliferator of Centrifuge designs.
Libya 2010 Squabbling over nuclear material
Libya 2011 Civil war
Fusion bombs use the reaction
Deuterium + Lithium6 --> Helium4 + Helium4 + EnergyUsing the "Isotopes and Atomic Mass" simulation at phet.colorado.edu, you can look up the mass of each nucleus. What fraction of the mass is converted to energy?
Z = Joules per kg generated by the fusion reaction W = Joules per nucleon generated by the fusion reactionDeuterium has 2 nucleons and Lithium6 has 6 nucleons, for a total of 8. What is Z and W?
Suppose a spaceship uses a rocket nozzle to channel the fusion products into a uni-directional flow. The maximum possible flow speed is
Z = .5 V^2What is V?
The maximum speed of a rocket is proportional to the exhaust velocity. If a rocket is moving at speed V, what is the time dilation factor?
Time dilation = (1-V^2/C^2)^(-1/2)Alpha Centauri is 4.4 light years away. How long would it take the rocket to get there?
A fusion rocket using Deuterium+Lithium6 is within our technological grasp. The only fusion reaction that delivers more energy is
Deuterium + Helium3 --> Helium4 + ProtonHelium3 is rare on the Earth but it can be found on the moon. An advanced civilizaton could potentially generate Helium3 through the reaction
Proton + Lithium6 --> Helium3 + Helium4Suppose a rocket uses the fission of Uranium-235. Using data from the web, what is W, Z, and V?